Research Article - (2024) Volume 15, Issue 3
Received: 05-Oct-2020, Manuscript No. JBSBE-23-8143;
Editor assigned: 08-Oct-2020, Pre QC No. P-8143;
Reviewed: 22-Oct-2020, QC No. Q-8143;
Revised: 19-Jul-2023, Manuscript No. R-8143;
Published:
16-Aug-2023
, DOI: 10.37421/2155-6210.2023.14.386
, QI Number: P-8143;
Citation: Jadhav, Dayanand B and Rajendra D Kokate.
"Green Synthesized Zinc Oxide Nanoparticles and Evaluation of their Use in
Nanoelectronics Gas Sensing Result and Performance." J Biosens Bioelectron 14 (2023): 386.
Copyright: �© 2023 Jadhav DB, et al. This is an open-access article distributed under the terms of the creative commons attribution license which permits
unrestricted use, distribution and reproduction in any medium, provided the original author and source are credited.
Highly stable, eco-friendly ZnO nanoparticles synthesized by an easy and cost-effective plant (Aloe barbadensis miller gel as crystal growth modifier) mediated synthesis route. The synthesized ZnO nanoparticles of average particle size 70 nm-90 nm. LPG sensing properties were systematically investigated. The ZnO nanoparticles exhibited outstanding gas sensing characteristics like higher gas response (~500 ppm LPG gas at 350°C), the reaction time (~3 sec), recovery time (~35 sec), excellent repeatability and good selectivity. Structural, morphological and phase conformation characteristics are measured using XRD, EDS, FESEM, TEM and FTIR. This work provides a completely unique green approach to synthesize ZnO nanoparticles for the LPG sensing application.
Eco-friendly • Nanoparticles • Cost-effective • LPG sensing application
The nanoparticles used in the various applications, having large surface to volume ratio. As per the estimate in all over world consumption of nanoparticles is growing from 225,060 metric tons to near about 585,000 metric tons between the year 2014-2019. Synthesis of the metal oxide nanoparticles occurred using different methods. The traditional chemical synthesis methods are metal reduction techniques, bottom-up methods and top-down methods. The metal reduction synthesis takes place with the help of reduction of metal ion soluble with the reducing or capping agents like sodium borohydride, ascorbic acid and hydrazine. The synthesis of metal nanoparticle takes place using the different methods i.e., using chemical and green synthesis methods [1].
The chemical synthesis method is expensive and unsafe chemicals used in order that nanomaterials occurred also toxic in nature. Biological synthesis routes using plant extracts provide a replacement and promising synthesis technique to synthesize several metal oxides nanoparticles, which are more eco-friendly and allows a controlled synthesis with well-defined size and shape of nanoparticles. During this article, the aloe vera plant is employed due to its stability synthesized zinc oxide nanoparticles characterized using XRD, FE-SEM, EDX and FT-IR.
Aloe-vera plant leaves contain a good range of bio-active compounds like glucose, vitamins, a molecule, minerals, cellulose fibers, salicylic acids and amino acids. Aloe-vera was chosen due to its functional properties like anti-inflammatory, antioxidant, antifungal and antibacterial. Furthermore, the importance of usage of natural, renewable and low-cost material, aloe-vera could ready to produce the metal oxide nanoparticles with an aqueous medium by avoiding the presence of the hazardous substances and toxic solvents [2].
In this research work synthesized zinc oxide nanoparticles using the aloe-vera plant extract which an angiosperm class is belong to the Malvaceae family distributed in India. The bark and leaves are used for pharmaceutical purposes. Facile synthesis of ZnO nanoparticles was performed without using any extensive chemicals or excess energy to analyze the role of the plant extract is displayed within the formation, stabilization of ZnO nanoparticles synthesis. The crystal structure and surface morphology characterized using X-Ray Diffraction (XRD) and Scanning Electron Microscopy (SEM). Sensing characteristics of the ZnO nanoparticles to LPG gas systematically investigated.
Aloe vera leaves were collected from IITB, Maharashtra, India. Zinc nitrate hexahydrate (Zn(NO3)2.6H2O) was purchased from Merck, India Pvt. Ltd. The de-ionized water was used throughout the reaction process [3].
Synthesis of zinc oxide nanoparticles
0.5 M of aqueous solutions Zn(NO3)2.6H2O was dissolved in 70 mL of double distilled water and well stirred with a stirrer. The fresh plant of the aloe vera was washed with water. The aloe vera plant extract of 50 mL was added within the Zn(NO3)2.6H2O. The solution prepared kept at 80°C during a petry dish and heated continuously until the water vaporized completely the obtained precursor thoroughly washed with distilled water several times to avoid K+ ion contamination and further dried in an oven at 70°C and apart of the as dried sample was calcinated within the air at 250°C for 2 hours to get the powder of ZnO nanoparticles [4].
Characterization
The sample was investigated by using X-Ray Diffraction (XRD) technique. The X-ray diffraction patterns were recorded with a Rigaku diffractometer having Cu KL (L=0.1542 nm).
Gas sensing study
The ZnO nanoparticles powder was wont to prepare thin films on the alumina substrate using spin coating technique and contacts were made with the help of silver paste to make the sensing element. The sensing observation administrated by using the ZnO detector within the gas chamber to sense Liquid Petroleum Gas (LPG). The detector had kept directly on a heater (chrome-alumni coil) within the death chamber and therefore the temperature was varied from 150°C to 350°C. Using digital multimeter the resistance is measured. The electric resistance of the detector was measured before and after exposure to focus on gas employing a sensitive digital multimeter. The performance of the detector is given as: The FESEM characterizations were performed using instrument model Zeiss/Ultra 55. The equipment FTIR with a category is litho/ analytical and make/model is Perkin Elmer/Spectra 100 (Serial number 83476) [5].
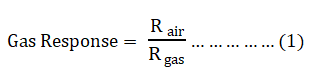
XRD results
The XRD patterns of as-prepared after annealing in air, the XRD pattern of ZnO after annealing and synthesized as shown in Figure 1. It exhibits the diffraction peaks like the ZnO (JCPDS No.: 043-0002). No other peaks were observed, indicating that no impurities were present and confirming that the adopted synthesis route gives pure ZnO [6].
The crystallite size was estimated by using the Scherrer formula:
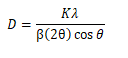
If the grains are spherical, k=0.9. λ=wavelength of X-ray radiation, β=peak Full Width at Half Maximum (FWHM). The crystalline size is to be calculated in the range of 75 nm-90 nm.
FT-IR spectroscopy
The FTIR analysis was evaluated to work out the functional groups within the ZnO nanoparticles. Figure 2 revealed the presence of the characteristic bands at ~526 cm-1, 675 cm-1, 1030 cm-1, 1395 cm-1 and 1586 cm-1 confirms its successful synthesis of shows that the FTIR spectra of ZnO nanoparticles within the 500 cm-1-3000 cm-1 regions. The broad peak 1395 cm-1 was indicated the OH stretching vibrations. The sharp peak present within the range of 1586 cm-1 indicates the free group [7].
Energy Dispersive X-ray spectroscopy (EDX)
The EDX spectra shows, that the ratio zinc and oxygen are such that the oxygen is less than the zinc which indicates that the ZnO synthesized is oxygen deficient as indicated in Figures 3 and 4.
Surface morphology
The FESEM images of ZnO nanoparticles formed after annealing (in air) shows that the nanoparticles of size 60 nm-90 nm are successfully synthesized using green approach as in Figure 5 [8].
LPG gas sensing characteristics
The effect of operating temperature on the gas response to 500 parts per million (ppm), LPG gas of prepared nanoparticles is shown in Figure 6. At 350°C, the gas response is peaked at its maximum value of 435. Above 350°C, the gas response decreased because the operating temperature increased further. From this result, it's concluded that optimum operating temperature for the green synthesized ZnO nanoparticle to detect LPG gas is at 350°C [9].
The characteristic shows the performance of the sensor. The recovery time is that the reverse process of response time when removal LPG gas as shown in Figure 7. After the introduction of LPG gas sensor elements responds quickly and recovers it's noted for ZnO the response time is near about 3 to 5 seconds and therefore the recovery time is near about 25 to 45 seconds. Sensitivity was measured as a change in resistance before and after exposure to focus on gas. Because the progressive adsorption and subsequent surface reactions occur with a rise within the temperature, the gas response of the sensor continuously increases because the temperature increases from 175°C to 350°C. At temperature 350°C, the optimum balances between the adsorption and desorption, the surface reactions and diffusion length could also be established and consequently, the LPG reacts most effectively with chemisorbed oxygen at such temperature, which ends up within the significant decrease within the resistance of the sensor.
Therefore, the utmost gas response of the ZnO nanoparticlesbased sensor towards LPG is predicted at such temperature. The reproducibility of the ZnO nanoparticles-based sensor was investigated by repeating the test 3 times. The representative dynamic gas response of the sensor upon periodic exposure to 500 ppm LPG at the optimum operating temperature of 350°C is shown in Figure 7. The sensor showed good reproducibility and reversibility upon repeated exposure and removal of LPG under same conditions. Furthermore, the repeated tests revealed that the gas response values are maintained and therefore the recovery abilities aren't reduced after several sensing cycles. Thus, the ZnO nanoparticles-based sensor exhibits a stable and repeatable characteristic, which suggests that it are often used as a reusable sensing material for the detection of LPG [10].
The sensing takes place by two methods increases within the current and reduces within the current. The present increases because of the reducing gases and therefore the current decreases thanks to oxidizing gases. During this method, current increases thanks to a decrease within the resistance thanks to the ZnO nanomaterial sample interact with the LPG gas molecules. The LPG gas contains propane and butane with a little amount of hydrocarbons. The formula for LPG is when propane (3) then C3H8 and when the butane (4) then C4H10. Therefore the generalized sort of LPG is CnH2n+2. It had been observed that the resistance of the sensing zinc oxide element increases when exposed to the LPG gas. The ZnO nanomaterial shows good reproducibility and reversibility upon periodic exposure of the gas about 500 ppm and removal of LPG gas under same conditions. During this way, the ZnO nanoparticles have good stability also as repeatability of the response for the target gas from Figures 8 and 9 [11].
A facile technique for the synthesis of metal oxides by the plantmediated synthesis method has been reported. This method offers a really simple, non-toxic, low cost, environment-friendly route for the synthesis of zinc oxide nanoparticles. The synthesized flowers of zinc nanoparticles were characterized by various characterization techniques like XRD, FE-SEM, FT-IR and EDX. The gas sensing properties of the sensor fabricated using the thin film of ZnO nanoparticles was measured by exposing it to different tested gases, including LPG, CO, CO2, H2 and ethanol at the operating temperature from 175°C to 400°C. It reveals that the sensor showed high sensitivity to 500 ppm LPG with the response nearly adequate to 521 at the optimal temperature of 350°C, which was a minimum of twice above that to other gases, implying the preferable selectivity of the sensor to LPG.
The ZnO nanoparticles were successfully synthesized by an easy plant-mediated synthesis method. It had been observed that the synthesized ZnO nanoparticles are highly oxygen deficient which significantly enhances the sensitivity of ZnO nanoparticles to LPG. The sensitivity to 500 ppm LPG is maximum at 350°C and it's found to be ~521.35. The reaction time and recovery time are nearly 2 s and 40 s respectively.
The authors acknowledge the facilities within the Indian nanoelectronics users program at IIT Bombay. This project approved by INUP IIT Bombay. The authors present sincere gratitude to director INUP IIT Bombay Dr. K. Nageshawari, project mentor INUP IITB Prof. Madhuri.
No conflict of interest was reported by the authors.
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
[Crossref] [Google Scholar] [PubMed]
Biosensors & Bioelectronics received 6207 citations as per Google Scholar report